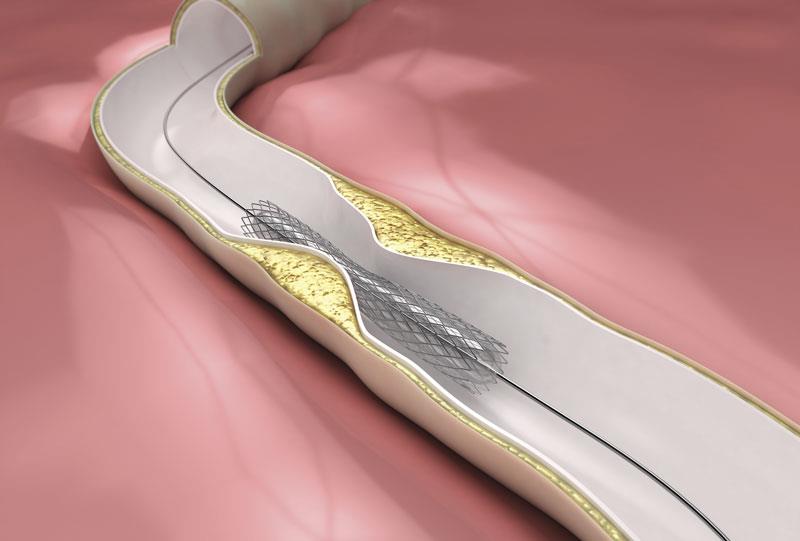
Several bioresorbable scaffolds are now available to further improve the convenience and outcomes of percutaneous coronary intervention (PCI) while enabling stent implantation in difficult cases.
“Bioresorbable scaffolds are the fourth revolution in interventional cardiology, coming after the invention of balloon angioplasty, bare metal stents and drug-eluting stents. It is the focus of coronary stent development today,” said Dr. Chung-Seung Chiang of the Division of Cardiology, Queen Elizabeth Hospital, Hong Kong. “The use of bioresorbable scaffolds is based on the assumption that there is only a transient need for blood vessel support. The scaffolds leave no permanent metallic implant in the body, allowing the restoration of natural vasomotion and future re-intervention.”
The first bioresorbable scaffold, Absorb, is an everolimus-eluting scaffold made of poly-L-lactide (PLLA). “It can be slowly metabolized and fully absorbed by the body in about 2 years,” explained Chiang.
The safety of Absorb was demonstrated in a small trial (n=99) presented at the American College of Cardiology’s 2014 Annual Scientific Session. “At 4 years, MI and hierarchical major adverse cardiac events [MACE] occurred in only 3 and 10.1 percent of patients, respectively, and 7.1 percent required target lesion revascularization. No cardiac death or scaffold thrombosis was observed,” reported Chiang. [J Am Coll Cardiol 2014;63(Suppl), doi: 10.1016/S0735-1097(14)61709-5] “Absorb was approved in Europe in 2011, but not yet by the US FDA. Several clinical trials are ongoing to assess its other benefits in different populations.”
Chiang also discussed some of the newer bioresorbable scaffolds with potential advantages over Absorb.
“The European-approved DESolve has a faster degradation of about 1 year and higher plasticity than Absorb,” said Chiang. “In its main trial of 123 patients, the 1-year clinical outcome was promising, with a low MACE rate of 5.69 percent and no stent thrombosis.” [Verheye S, TCT 2013]
“DREAMS, a drug-eluting absorbable metal scaffold, is made of magnesium alloy and PLLA. The device is thinner than Absorb [120 µm vs 157 µm], permitting easier delivery to the site of lesion,” he said. “Its safety and efficacy are currently being investigated in the BIOSOLVE-II study.”
“Another investigational scaffold is the desaminotyrosine polycarbonate polymer-based ReZolve2, which has a higher radial strength than Absorb and is easily trackable with its exclusive visibility under X-ray,” Chiang noted.